Parameter
Potential Technology
Isobutane Content (%)
Process Gas Chromatograph (GC)
Acid Strength (98-99%)
Density Meter + Titration
Temperature Profile
Infrared Thermography
Hydrocarbon-in-Acid
Moisture (ppm)
Tunable Diode Laser (TDLAS)
Water Content, Hydrocarbons
NIR Spectroscopy
i-C4 Purity
FT-NIR Analyzer
RON, MON, Sulfur
Octane Analyzer + XRF
pH, Oil Content
Electrochemical pH + UV-Vis
Description
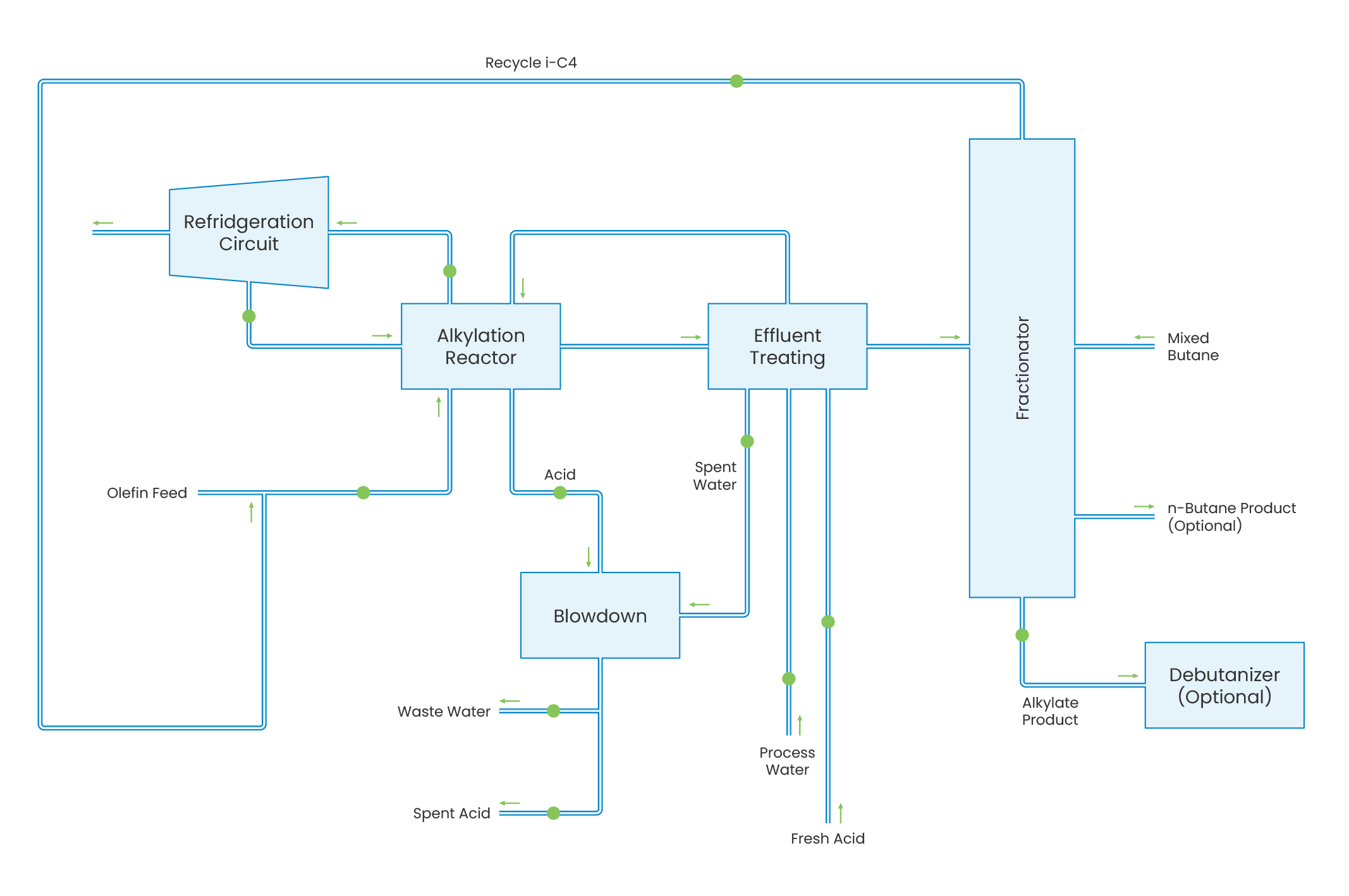
An alkylation unit is designed to combine isobutane with C4 olefins in the presence of a strong acid catalyst. The resulting highly branched, paraffinic product is a low vapour pressure, high octane blending component. The olefin feed is initially combined with recycle isobutane from the fractionation and then cooled by feed/effluent heat exchange. This combined stream is fed to the contactor reactors, where it is mixed with sulphuric acid catalyst to form an acid emulsion for alkylation of the olefins and isobutane. Note: Not all Alkylation processes uses sulphuric acid as the catalysing fluid, for example hydrofluoric can be used instead. Conversion of olefins to alkylate is essentially 100%. The hydrocarbon phase (reactor effluent) is separated from the acid emulsion in the acid settlers. Acid-rich emulsion is recycled to the contactor reactor.The net effluent stream from the reaction section contains traces of free acid, alkyl sulphates and di-alkyl sulphates which can cause corrosion and fouling in downstream and is therefore treated to remove these. The treated effluent is then passed on to the fractionation section to separate the isobutane for recycle to the reactors, the n-butane fraction for recycle to the C4 Isomerisation Unit and the alkylate product for blending in the gasoline pool.