H₂S/SO₂ Ratio (2:1)
COS/CS₂
- H₂S
- SO₂
- Sₓ Vapour
- H₂S
- SO₂
- Sₓ Vapour
- H₂S
- SO₂
- Sₓ Vapour
- H₂S (<0.5 vol%)
- Liquid Sulfur Purity
- H₂S (<0.5 vol%)
- Liquid Sulfur Purity
- H₂S (<0.5 vol%)
- Liquid Sulfur Purity
- Total Sulfur (H₂S+SO₂)
- SO₂ (ppm)
- H₂S (dissolved)
- Purity (99.9%)
- H₂S (dissolved)
- Purity (99.9%)
- H₂S (dissolved)
- Purity (99.9%)
- H₂S (dissolved)
- Purity (99.9%)
- Liquid Hydrocarbons (BTEX)
- H₂S
- H₂O carryover
- NH₃ slip
H₂S/SO₂ Ratio (2:1)
COS/CS₂
- H₂S
- SO₂
- Sₓ Vapour
- H₂S
- SO₂
- Sₓ Vapour
- H₂S
- SO₂
- Sₓ Vapour
- H₂S (<0.5 vol%)
- Liquid Sulfur Purity
- H₂S (<0.5 vol%)
- Liquid Sulfur Purity
- H₂S (<0.5 vol%)
- Liquid Sulfur Purity
- Total Sulfur (H₂S+SO₂)
- SO₂ (ppm)
- H₂S (dissolved)
- Purity (99.9%)
- H₂S (dissolved)
- Purity (99.9%)
- H₂S (dissolved)
- Purity (99.9%)
- H₂S (dissolved)
- Purity (99.9%)
- Liquid Hydrocarbons (BTEX)
- H₂S
- H₂O carryover
- NH₃ slip
Parameter
Potential Technology
H₂S/SO₂ Ratio (2:1)
UV-DOAS
COS/CS₂
FT-IR
H₂S, SO₂, Sₓ Vapour
Laser Spectroscopy (TDLAS)
H₂S (<0.5 vol%)
Electrochemical Cell
Liquid Sulfur Purity
X-ray Fluorescence (XRF)
Total Sulfur (H₂S+SO₂)
UV Fluorescence
SO₂ (ppm)
CEMS (NDIR+UV)
H₂S (dissolved)
Colorimetric Analyzer
Purity (99.9%)
NIR Spectroscopy
Liquid Hydrocarbons (BTEX), H₂S
Near-Infrared (NIR) Analyzer
H₂O carryover, NH₃ slip
Moisture Analyzer + Laser NH₃
Parameter
H₂S (<0.5 vol%)
Liquid Sulfur Purity
Potential Technology
Electrochemical Cell
X-ray Fluorescence (XRF)
Parameter
Total Sulfur (H₂S+SO₂)
SO₂ (ppm)
Potential Technology
UV Fluorescence
CEMS (NDIR+UV)
Parameter
H₂S (dissolved)
Purity (99.9%)
Potential Technology
Colorimetric Analyzer
NIR Spectroscopy
Parameter
Liquid Hydrocarbons (BTEX), H₂S
Potential Technology
Near-Infrared (NIR) Analyzer
Description
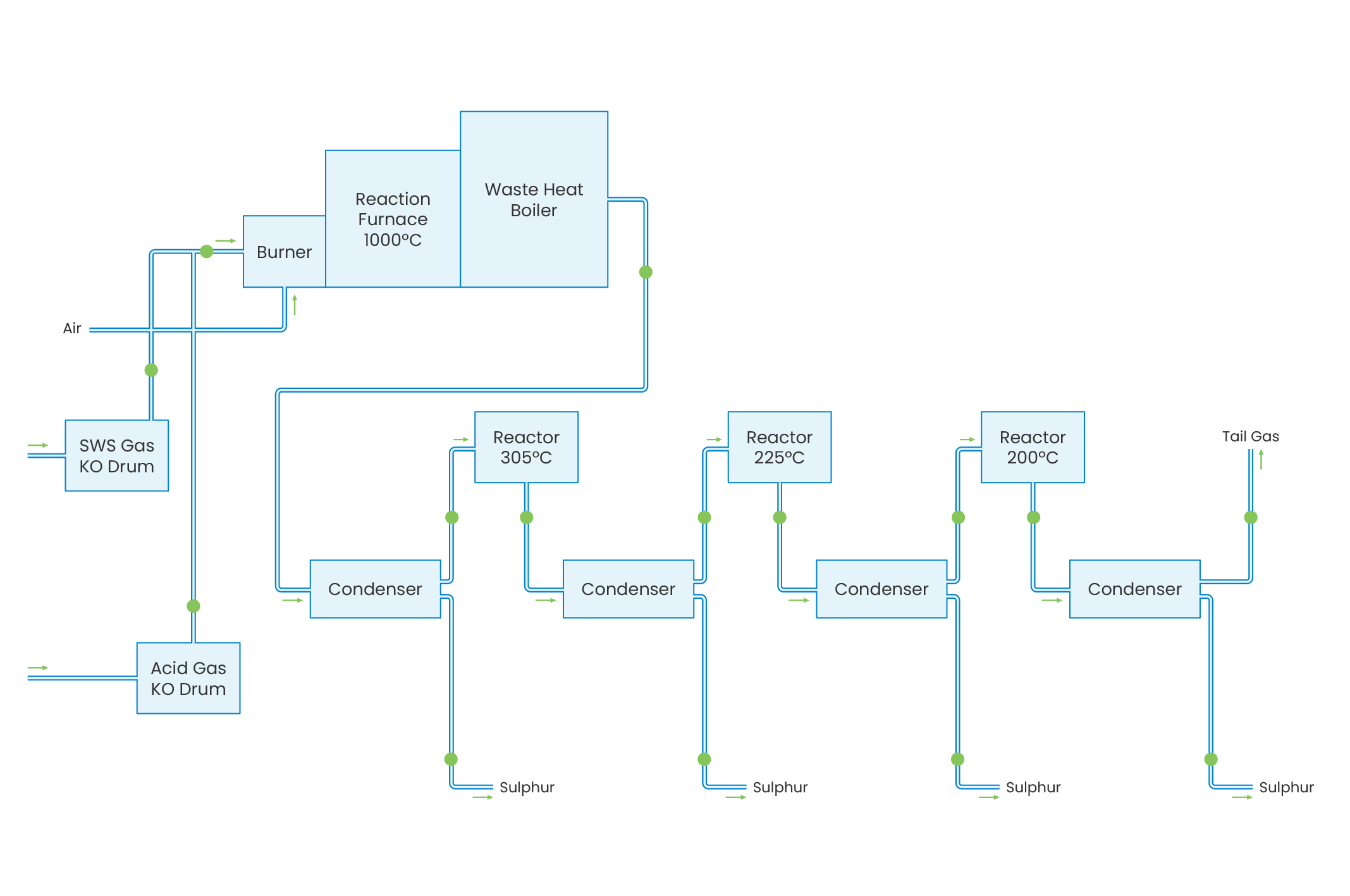
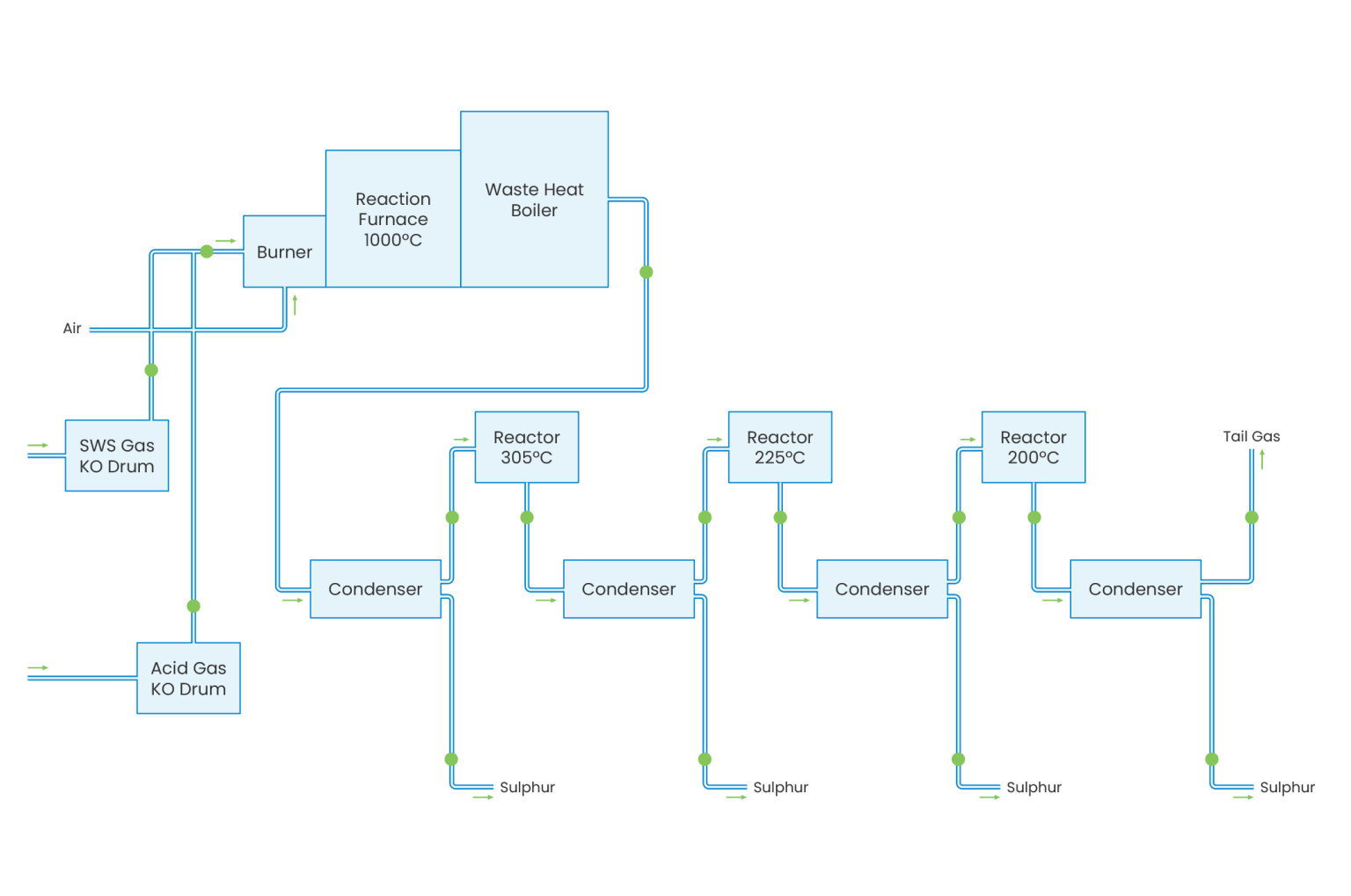
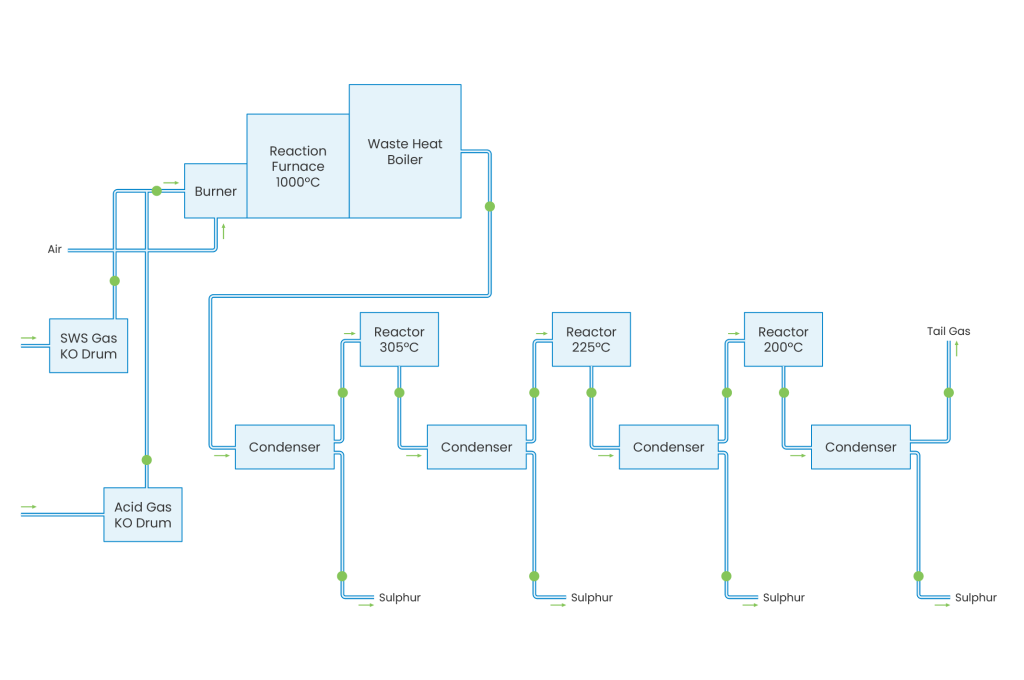
The Sulphur Recover Unit (SRU) or Claus Process has been developed to recover elemental sulphur from H2S/NH3-containing gases originating from gas treating plants such as Sour Water Strippers. There are two main varieties of the Claus process: straight through and split flow. The straight through process is used for feed gas containing more than 50% H2S, while split flow is applied for gases containing 50 to 15% H2S. The sulphur recovery process consists of a thermal stage (combustion chamber, waste heat boiler) and two or three catalytic reaction stages (reheater, reactor and condenser).
The Claus process is based on the combustion of part of the H2S to SO2 and the subsequent reaction between the remaining H2S and the SO2 produced, forming sulphur. In the thermal stage, the Claus reaction takes place at a high temperature level (950-1350°C). The sulphur produced in this stage is condensed either in the waste heat boiler or in the condenser. Subsequently, the gas is reheated to a temperature of 200-300°C before introduction into each reactor stage. The heat generated from the reactions is used for low, medium, or high steam production. In the sulphur condensers low pressure steam is generated. The tail gas is sent to an incinerator or tail gas treater.
Depending on the number of reactors and the quality of the feed gas, sulphur recoveries may vary from 95 to 98% weight recovery. To increase the sulphur recovery, the tail gas is sent to the Tail Gas Treatment Unit (TGTU), which removes the remaining sulphur by a combination of chemical reaction and absorption. Incinerated tail gas is discharged to the atmosphere through an elevated stack.
